A total internal reflection fluorescence microscope (TIRFM) is a type of microscope with which a thin region of a specimen, usually less than 200 nanometers can be observed. Source: https://en.wikipedia.org/wiki/Total_internal_reflection_fluorescence_microscope
Background
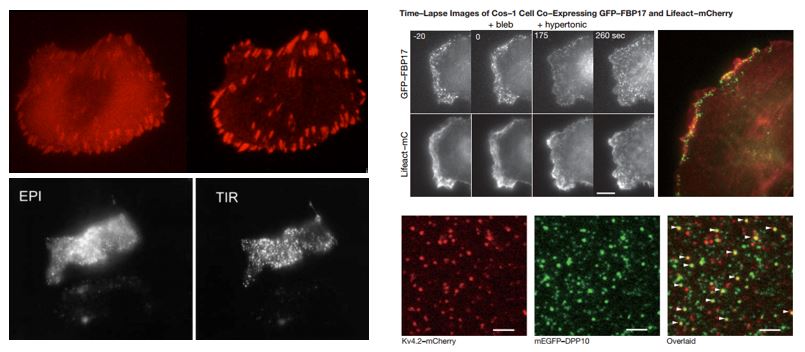
In cell and molecular biology, a large number of molecular events in cellular surfaces such as cell adhesion, binding of cells by hormones, secretion of neurotransmitters, and membrane dynamics have been studied with conventional fluorescence microscopes. However, fluorophores that are bound to the specimen surface and those in the surrounding medium exist in an equilibrium state. When these molecules are excited and detected with a conventional fluorescence microscope, the resulting fluorescence from those fluorophores bound to the surface is often overwhelmed by the background fluorescence due to the much larger population of non-bound molecules. TIRFM allows for selective excitation of the surface-bound fluorophores, while non-bound molecules are not excited and do not fluoresce. Due to the fact of sub-micron surface selectivity, TIRFM has become a method of choice for single molecule detection.
Overview
The idea of using total internal reflection to illuminate cells contacting the surface of glass was first described by E.J. Ambrose in 1956.[1] This idea was then extended by Daniel Axelrod[2] at the University of Michigan, Ann Arbor in the early 1980s as TIRFM. A TIRFM uses an evanescent wave to selectively illuminate and excite fluorophores in a restricted region of the specimen immediately adjacent to the glass-water interface. The evanescent wave is generated only when the incident light is totally internally reflected at the glass-water interface. The evanescent electromagnetic field decays exponentially from the interface, and thus penetrates to a depth of only approximately 100 nm into the sample medium. Thus the TIRFM enables a selective visualization of surface regions such as the basal plasma membrane (which are about 7.5 nm thick) of cells as shown in the figure above. Note, however, that the region visualized is at least a few hundred nanometers wide, so the cytoplasmic zone immediately beneath the plasma membrane is necessarily visualized in addition to the plasma membrane during TIRF microscopy. The selective visualization of the plasma membrane renders the features and events on the plasma membrane in living cells with high axial resolution.
TIRF can also be used to observe the fluorescence of a single molecule,[3][4] making it an important tool of biophysics and quantitative biology.
Cis-geometry (through-objective TIRFM) and trans-geometry (prism- and lightguide based TIRFM) have been shown to provide different quality of the effect of total internal reflection. In the case of trans-geometry, the excitation lightpath and the emission channel are separated, while in the case of objective-type TIRFM they share the objective and other optical elements of the microscope. Prism-based geometry was shown to generate clean evanescent wave, which exponential decay is close to theoretically predicted function.[5] In the case of objective-based TIRFM, however, the evanescent wave is contaminated with intense stray light. The intensity of stray light was shown to amount 10-15% of the evanescent wave, which makes it difficult to interpret data obtained by objective-type TIRFM [6][7]
Source: https://en.wikipedia.org/wiki/Total_internal_reflection_fluorescence_microscope
Compared with the traditional objective type Point-TIRF, the laser is focused to the edge of the back focal plane of the high NA objective. The incident light illuminates the sample from one side of the objective and reflects back to the objective from the other side. The laser beams on the sample and container. The 200nm deep evanescent field is generated at the bottom interface, which can excite the fluorescence signal of the sample in the evanescent field. The 360°Ring TIRF is switched at a high speed by a set of XY scanning galvanometers, so as to produce a ring illumination on the back focal plane of the objective. The incident light can be irradiated to the sample from multiple directions at the same time, thereby generating a more uniform evanescent field and eliminating Interference fringes during laser illumination. At the same time, the 360°Ring TIRF can easily switch the illumination depth of the evanescent field. By dragging the angle slider, the evanescent field depth of different excitation wavelengths can be optimized.
The 360°Ring TIRF system consists of Obrital TIRF optical path module (including fiber interface, laser collimation, XY scanning galvanometer and focusing lens), laser coupler unit VS-LMS (including 405/488/561/640 solid-state laser, Lasers coupling, single-mode fiber, laser switch and intensity module) and VisiView TIRF imaging software, which can be mounted on a research-grade inverted fluorescence microscope and use SCMOS camera (Prime 95B) for single-molecule fluorescence imaging.